Research and Application of Ultraviolet (UV) Curing Adhesives
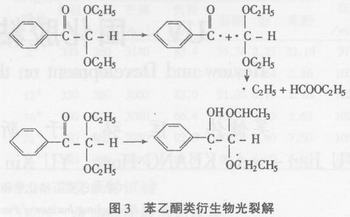
The rate of initiation of dialkoxyacetophenones is faster than that of benzoin alkyl ethers. This may be due to the secondary cleavage of the free-radical and free radical-coincidence reactions produced by their photolysis, resulting in highly reactive alkyl radicals. The reason.
Some common cracking UV photoinitiators molecular formula and UV absorption characteristics shown in Figure 4

Among them, Lucirin TPO and Irgacure 369 are reported to have higher light efficiency and better overall performance.
In the use of photoinitiators, amines are sometimes added as sensitizers. The mechanism is based on the analysis of the formation of amino radicals after the absorption of oxygen atoms, reducing the inhibition of oxygen on the free radical curing reaction.
Cracking photoinitiators are not completely consumed during curing and sometimes have low molecular weight residues. This is detrimental to the performance of the cured adhesive. Recently, charge transfer type photocuring agents have attracted attention.
The curing mechanism is shown in Figure 5:

Figure 5 Photopolymerization initiated by charge transfer ligands
The advantage of this kind of curing agent is that there is no low molecular residue. The charge supplier is a maleimide (MI) derivative and the charge acceptor is an alkyl vinyl ether. Presently reported curing agents of this type are mostly used in alkyl propenyl ether systems, and cure speed is not too fast.
(to be continued)
White Cashmere Top,Grey Cashmere Top,Brown Cashmere Top
Cashmere Sweater,Cashmere Blanket Co., Ltd. , http://www.nscashmeresweater.com